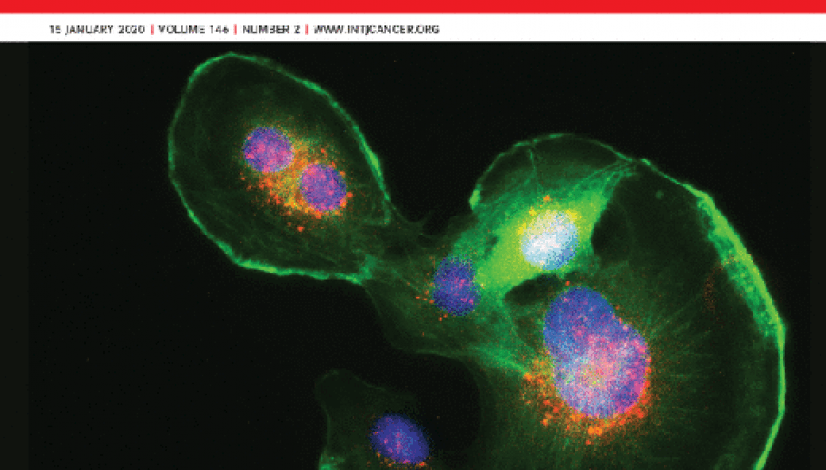
Epigenetic downregulation of TET3 reduces genome-wide 5hmC levels and promotes glioblastoma tumorigenesis
Detalles de la Publicación
Revista: Int J Cancer. 2020 Jan 15;146 (2): 373-387
Autores: Antonella Carella, Juan R. Tejedor, María G. García, Rocío G. Urdinguio, Gustavo F. Bayón, Marta Sierra, Virginia López, Estela García-Toraño, Pablo Santamarina-Ojeda, Raúl F. Pérez, Timothée Bigot, Cristina Mangas, María D. Corte-Torres, Inés Sáenz-de-Santa-María, Manuela Mollejo, Bárbara Meléndez, Aurora Astudillo, María D. Chiara, Agustín F. Fernández, Mario F. Fraga
Factor de Impacto: 4,892
Resumen
Loss of 5-hydroxymethylcytosine (5hmC) has been associated with mutations of the ten–eleven translocation (TET) enzymes in
several types of cancer. However, tumors with wild-type TET genes can also display low 5hmC levels, suggesting that other
mechanisms involved in gene regulation might be implicated in the decline of this epigenetic mark. Here we show that DNA
hypermethylation and loss of DNA hydroxymethylation, as well as a marked reduction of activating histone marks in the TET3
gene, impair TET3 expression and lead to a genome-wide reduction in 5hmC levels in glioma samples and cancer cell lines.
Epigenetic drugs increased expression of TET3 in glioblastoma cells and ectopic overexpression of TET3 impaired in vitro cell
growth and markedly reduced tumor formation in immunodeficient mice models. TET3 overexpression partially restored the
genome-wide patterns of 5hmC characteristic of control brain samples in glioblastoma cell lines, while elevated TET3 mRNA
levels were correlated with better prognosis in glioma samples. Our results suggest that epigenetic repression of TET3 might
promote glioblastoma tumorigenesis through the genome-wide alteration of 5hmC.